Ionic compounds undergo fast reactions in their aqueous solution. On passing an electric current through fused, molten and aqueous solution of electrovalent compounds, the ions dissociate and migrate towards electrodes.ħ. But in organic solvents like Phenol, as the dielectric constant is minimum, it increases the force of attraction making the ions more strongly held by the electrostatic force which makes the compounds insoluble.Ħ. Ionic Compounds are Soluble in Water but Insoluble in Organic Solvents like Benzene and Phenol.Īs water has maximum dielectric constant, therefore it decreases the force of attraction between the ions and thus it forms free ions and hence they dissolve in water. Ionic Compounds Act as Strong Electrolytes.Īs on dissolving in water, ionic compounds allow the passage of electric current through them due to the presence of free ions.ĥ. In aqueous solution, the high dielectric constant overcomes the strong electrostatic force of attraction, thus making the ions free to conduct electric current.Ĥ. But infused or molten state, these forces of attraction get weakened and thus the ions become free to conduct electricity. In solid-state, they do not conduct electricity as the ions are not free but held by the strong electrostatic force. However, they can conduct electricity in their fused, molten and in their aqueous solution. Ionic Compounds Do Not Conduct Electricity in Their Solid-State. As in these compounds, ions are held by the strong electrostatic force of attraction, so a large amount of energy is required to overcome these forces of attraction between the ions.ģ. 
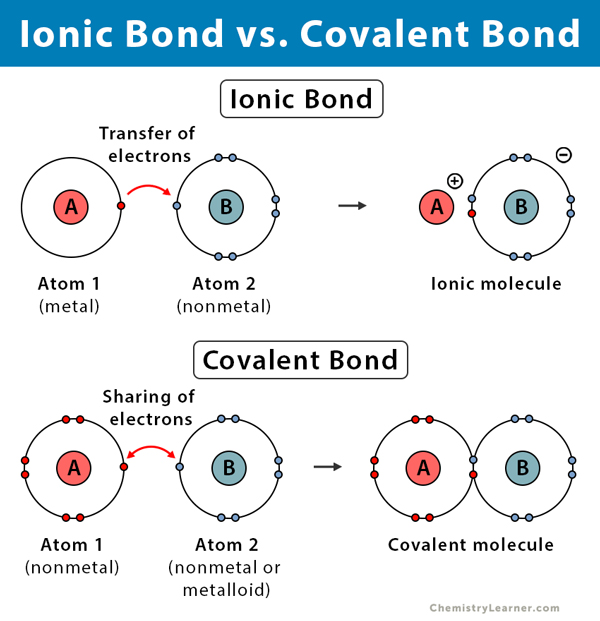
Ionic Compounds Have A High Melting Point and High Boiling Point. This is because their constituent particles are ions which are held by the strong electrostatic force of attraction and hence they cannot be separated easily.Ģ. What are the Properties of Ionic or Electrovalent Compounds? The chemical compounds formed as a result of the transfer of electrons from the outermost shell of metal to the outermost shell of a non-metal are called electrovalent compounds. And finally, electron affinity is defined as the energy released in addition of an electron to the outermost shell of an isolated gaseous atom. Ionization Potential is the energy required to remove an electron from the outermost shell of an isolated gaseous atom. the non-metals participating in an ionic bond formation should have high Electron Affinity. the metals participating in an ionic bond formation should have low Ionization Potential and 2. There are two essential factors for Ionic Bond formation: 1. The bond formed as a result of the transference of electrons from the outermost shell of metal to the outermost shell of a non-metal is alternatively known as an electrovalent bond. An ionic bond is defined as that bond between a metal and a non-metal which is responsible to hold the oppositely charged ions by the strong electrostatic force of attraction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
